In supercritical fluid (SCF) injection for footwear midsoles, nitrogen (N₂) is the dominant physical blowing agent used in commercial production. Carbon dioxide (CO₂) is also well understood from a materials science perspective and widely used in other foaming processes, but its use in SCF injection remains primarily in development. The fundamental difference between these gases lies in how they interact with polymer systems: N₂ provides process stability and uniform cell structure, while CO₂ offers higher solubility and expansion potential through plasticization effects. Mixed-gas systems aim to combine these behaviors, but are not yet widely implemented in production.
Unlike autoclave or chemical foaming processes, SCF injection is a fully thermoplastic process. It does not involve chemical blowing agents or crosslinking. Foam formation is driven entirely by dissolved gas and controlled pressure reduction during injection and molding, which places strict requirements on both material behavior and process control.
Fundamental Gas Property Differences (N₂ vs CO₂)
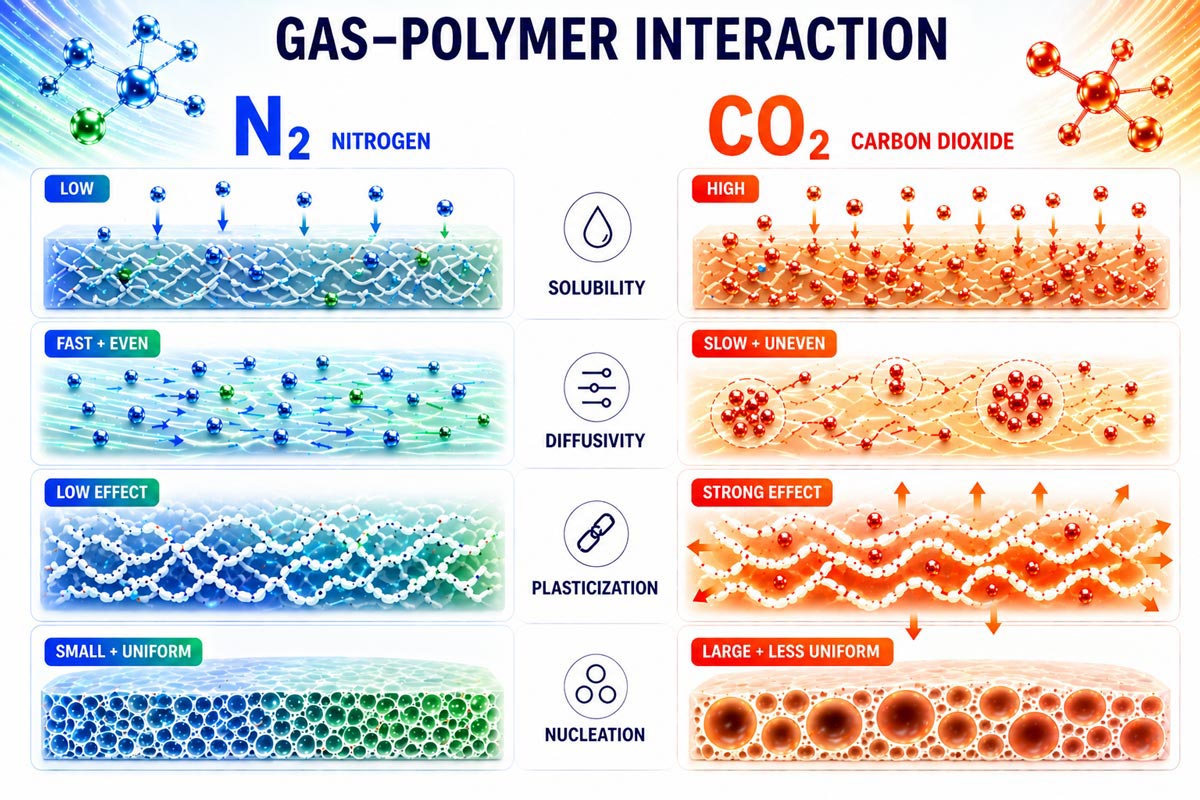
Solubility
One of the most important differences between nitrogen and carbon dioxide is solubility. CO₂ dissolves into most polymer systems at significantly higher levels than N₂, often several times greater depending on the material and processing conditions. This higher solubility allows for increased gas loading, which directly translates to greater expansion potential and the ability to achieve lower-density foam structures.
In contrast, nitrogen has relatively low solubility. While this limits maximum expansion, it provides a more controlled and predictable foaming process, which is critical for stable injection molding operations.
Diffusivity
Nitrogen typically exhibits higher diffusivity in polymer melts than CO₂, meaning it distributes more quickly and uniformly throughout the material prior to nucleation. This uniform distribution contributes to consistent cell structure and repeatable processing behavior.
CO₂ diffuses more slowly on its own, but its plasticization effect increases polymer chain mobility, which can enhance gas transport under certain conditions. Even with this effect, CO₂ systems generally require tighter control to ensure uniform gas distribution.
Plasticization Effects (CO₂-Specific)
A key distinguishing feature of CO₂ is its ability to plasticize polymers. When dissolved into the melt, CO₂ reduces viscosity and can lower the effective glass transition temperature of the material. This increases chain mobility, making it easier for the material to expand during pressure reduction.
While this behavior can be beneficial for achieving lower density, it also reduces melt strength. This creates a trade-off: CO₂ can enable greater expansion, but makes it more difficult to maintain structural stability during foam formation.
Nucleation Behavior
The choice of gas also influences nucleation behavior and resulting cell structure. Nitrogen tends to promote higher nucleation density, resulting in smaller and more uniform cells. This contributes to improved structural consistency and surface quality.
Carbon dioxide, due to its higher solubility and plasticization effects, generally produces lower nucleation density with larger cell structures. This supports greater expansion but can lead to less uniform morphology if not carefully controlled.
Mixed gas systems offer the potential to tune this balance, allowing for controlled adjustment of nucleation density and cell growth behavior.
SCF Injection Process Behavior
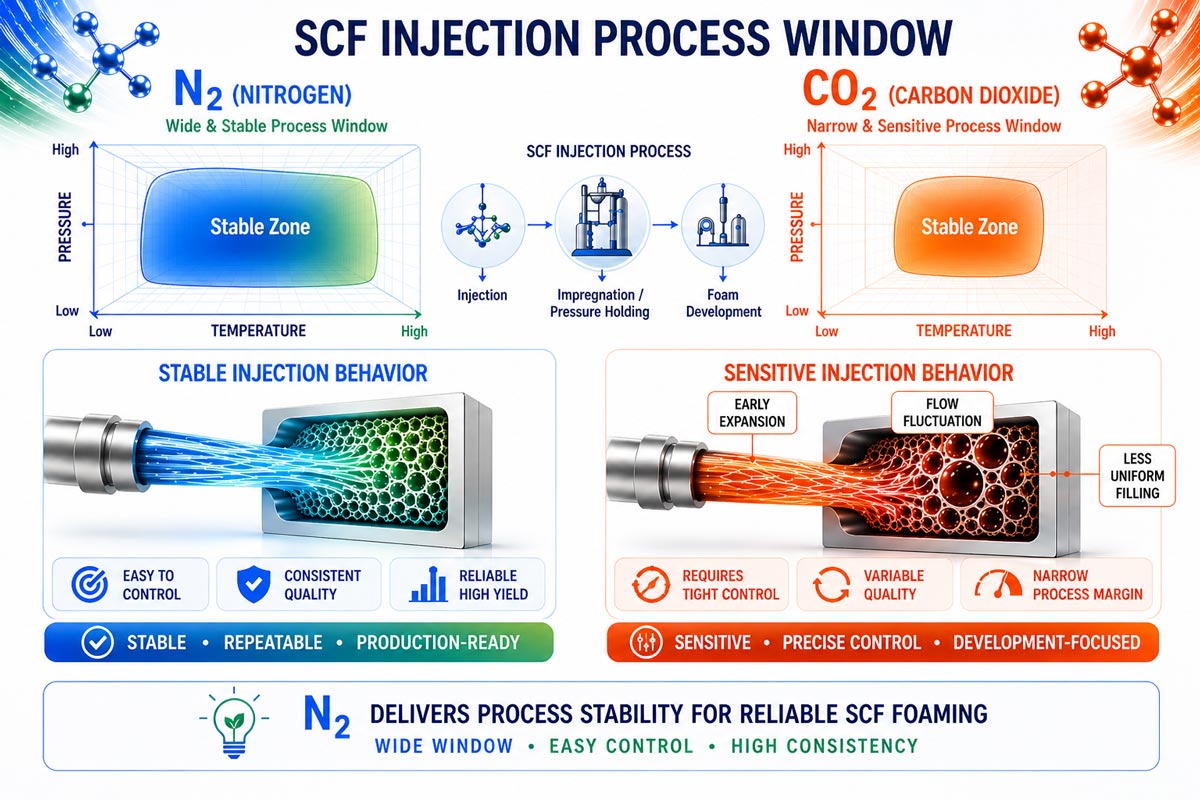
Gas Dissolution and Process Sensitivity
In SCF injection systems, nitrogen dissolves into the polymer at lower concentrations but does so in a stable and predictable manner. This contributes to a wide and forgiving process window, which is one of the key reasons it is used in all mainstream SCF injection systems currently in mass production.
Carbon dioxide, on the other hand, can dissolve at much higher concentrations. While this increases expansion potential, it also introduces greater sensitivity to temperature, pressure, and residence time. Small variations in processing conditions can have a larger impact on foam behavior.
Behavior During Injection and Pressure Drop
During injection and subsequent pressure reduction, nitrogen-based systems tend to nucleate in a controlled manner, enabling stable mold filling and consistent part formation. This predictability is critical in high-volume manufacturing.
CO₂-based systems exhibit stronger expansion tendencies due to higher gas loading and reduced melt viscosity. As a result, the timing of nucleation and cell growth becomes more sensitive, requiring tighter control of the process to avoid instability.
Process Stability and Control
From a production standpoint, nitrogen provides a more robust and stable processing window. This is why all mainstream SCF injection systems in commercial footwear production today rely on N₂ as the blowing agent.
CO₂ introduces a narrower processing window due to its combined effects on solubility, diffusivity, and melt strength. While these characteristics offer potential advantages, they also increase process complexity. Mixed gas systems can theoretically balance these effects but are not yet standardized in production environments.
Foam Structure and Performance Differences
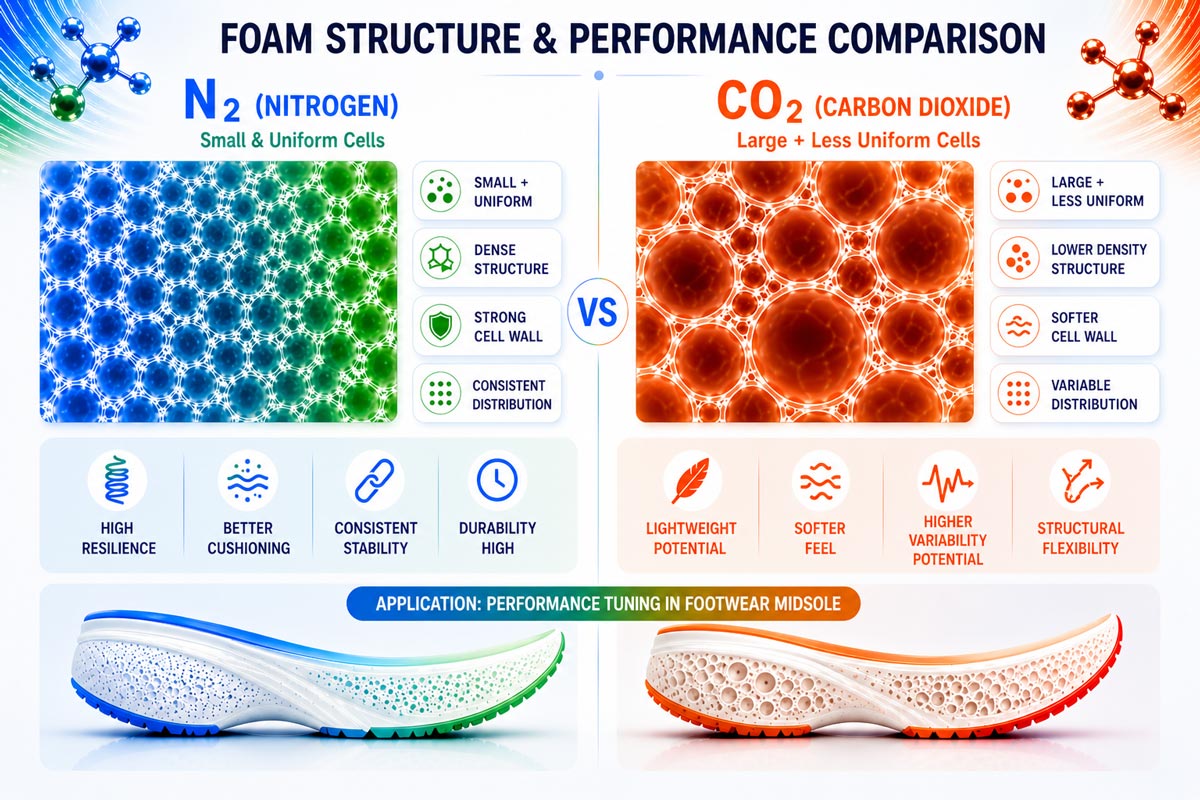
Density Capability
Carbon dioxide enables higher expansion potential due to its greater solubility, making it theoretically capable of achieving lower-density foam structures than nitrogen. However, achieving this consistently in an injection process remains challenging.
Nitrogen provides more controlled and repeatable density outcomes, even if the minimum achievable density is somewhat higher.
Cell Morphology
Nitrogen typically produces fine, uniform cell structures with high nucleation density. This leads to improved consistency and surface quality.
CO₂ tends to produce larger cells due to lower nucleation density and more aggressive expansion behavior. While this can reduce density, it can also introduce variability if not carefully controlled.
Mechanical Performance
Foams produced with nitrogen generally exhibit more consistent mechanical properties, including durability and structural integrity. This is a direct result of uniform cell structure and stable processing conditions.
CO₂-based foams may achieve lower weight and a softer feel, but can be more sensitive to structural inconsistencies. Mixed gas systems offer the potential to tune these properties, but are still under development in injection-based applications.
Practical Pros and Cons
N₂ (Nitrogen)
Advantages:
- Stable and well-understood process
- Widely used in commercial SCF injection
- Produces fine, uniform cell structures
- Broad and robust processing window
Limitations:
- Lower expansion potential compared to CO₂
CO₂ (Carbon Dioxide)
Advantages:
- High solubility enables greater expansion
- Plasticization improves flow and diffusion
- Potential for lower-density structures
Limitations:
- Use in SCF injection remains primarily in development
- Narrower and more sensitive process window
- Reduced melt strength during foaming
Mixed Gas Systems (N₂ + CO₂)
Advantages:
- Ability to tune nucleation and expansion behavior
- Potential to balance structure and density
Limitations:
- Increased process complexity
- Limited use in commercial production today
Practical Selection Guide
Material and production priorities ultimately determine gas selection:
- Use N₂ when stability, consistency, and scalability are required, particularly in commercial production environments and with established materials such as TPU and TPEE.
- Explore CO₂ in development settings when targeting lower density or enhanced expansion, especially when evaluating new material systems.
- Consider mixed gas approaches when fine-tuning foam structure and performance, recognizing that these systems require more advanced control and are not yet widely adopted.
Relationship to Autoclave Foaming
Carbon dioxide is widely used in autoclave foaming systems due to its plasticization effect and high solubility, which support gas diffusion over longer processing times. In these systems, CO₂ helps saturate the material prior to expansion.
SCF injection differs significantly. It is a fast, thermoplastic process without crosslinking, requiring precise control of gas dissolution, nucleation, and expansion within a much shorter cycle. These constraints make nitrogen more suitable for current injection-based manufacturing.
Future Outlook
Ongoing development in SCF injection foaming is focused on improving the interaction between gas systems and polymer materials. This includes exploration of mixed gas systems, improved material formulations, and more advanced gas dosing and control technologies.
As these developments progress, the role of CO₂ and hybrid gas systems may expand, particularly in applications targeting lower density and higher performance.
Q&A
Q: If CO₂ offers higher expansion potential, why isn’t it used in commercial SCF injection today?
A: CO₂ introduces greater process sensitivity due to its high solubility and plasticization effects, making it more difficult to control during injection. Nitrogen provides a more stable and reliable process window for production.
Q: What makes nitrogen the standard gas for SCF injection systems?
A: Nitrogen offers consistent dissolution, high diffusivity, and controlled nucleation behavior, resulting in stable processing and repeatable foam structures at production scale.
Q: Can mixed gas systems outperform pure nitrogen systems?
A: Mixed gas systems have the potential to balance nucleation and expansion behavior, but they introduce additional complexity and are still primarily used in development rather than large-scale production.
Q: How does CO₂ change polymer behavior during SCF injection?
A: CO₂ plasticizes the polymer, reducing viscosity and increasing chain mobility. This enhances expansion but reduces melt strength, making the process more sensitive to control.
Q: Does SCF injection use the same gas strategies as autoclave foaming?
A: No. Autoclave systems commonly use CO₂ due to longer processing times and different mechanisms, while SCF injection relies primarily on nitrogen for stability and control.
Note
Gas behavior, foam structure, and performance are highly dependent on polymer type, processing conditions, and equipment design. As SCF injection technology evolves, the role of CO₂ and mixed gas systems may expand.