The intersection of food processing and materials engineering often yields unexpected innovation pathways. One such convergence exists between the decaffeination of coffee using supercritical carbon dioxide and the development of high-performance foams in footwear applications. Although the underlying scientific principles are shared, the domains of application span from organic compound extraction to mechanical performance tuning in polymeric systems. This article explores the parallels between these two applications, focusing on the role of supercritical fluids (SCFs) in modulating molecular interactions, phase behavior, and material morphology.
Supercritical Carbon Dioxide in Coffee Decaffeination
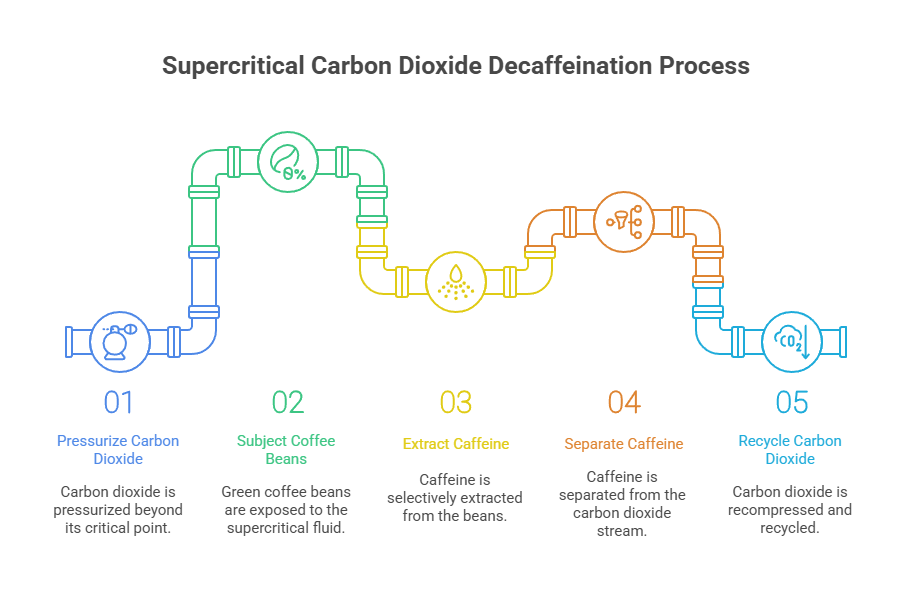
The decaffeination process utilizing supercritical carbon dioxide is a solvent-based extraction that targets caffeine molecules while minimizing the loss of desirable volatile compounds responsible for flavor and aroma. In this process, green coffee beans are subjected to pressurized carbon dioxide beyond its critical point, typically at pressures exceeding 73 atmospheres and temperatures above 31 degrees Celsius. The supercritical fluid exhibits gas-like diffusivity and liquid-like solvation capabilities, enabling it to penetrate deep into the porous structure of the coffee bean matrix.
Caffeine, being moderately nonpolar and relatively small in molecular weight, exhibits high solubility in supercritical carbon dioxide. The process is further optimized by co-solvent modifiers such as water or ethanol, which adjust the polarity of the SCF phase to increase caffeine selectivity. Once solubilized, caffeine is separated from the SCF stream via pressure reduction or adsorption techniques. The carbon dioxide is then recompressed and recycled, allowing for continuous operation with high energy efficiency and minimal environmental impact.
The success of this process hinges on the tunability of SCFs. By modulating pressure and temperature, the solvating power of the fluid can be fine-tuned to selectively extract compounds based on their molecular weight, polarity, and diffusivity. These thermodynamic control mechanisms are directly translatable to polymeric systems, especially those used in high-performance foams.
SCF Processing in Footwear Foam Fabrication
In the performance footwear industry, material innovation increasingly targets lightweight structures, enhanced energy return, and controlled compression response. Supercritical fluid foaming techniques, particularly those utilizing supercritical carbon dioxide or nitrogen, have emerged as a transformative platform for manufacturing polymeric foams with engineered microstructures.
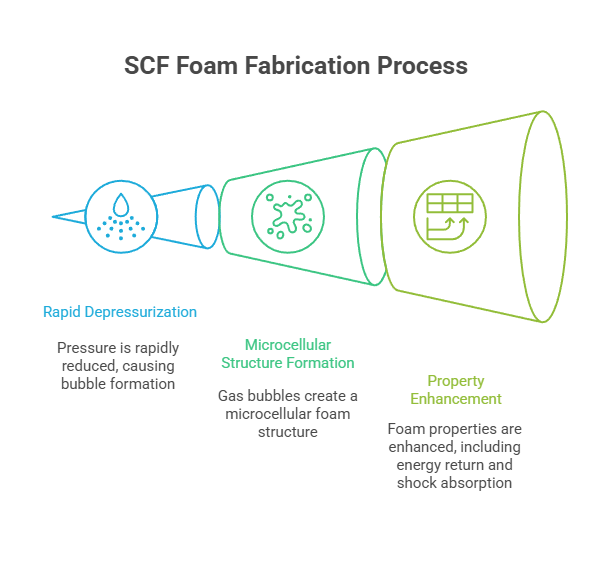
The core concept involves saturating a polymer melt or solid preform with a supercritical fluid under controlled temperature and pressure. Upon rapid depressurization, the dissolved SCF nucleates into gas bubbles, forming a microcellular structure. The resulting foam’s morphology, including cell size, density, and distribution, is highly sensitive to the SCF process parameters.
Much like the caffeine extraction process, the SCF’s behavior is manipulated to induce specific physicochemical changes in the host matrix. In foam processing, this tunability is exploited to control nucleation kinetics, polymer chain mobility, and gas-polymer interfacial tension. For instance, a higher saturation pressure generally leads to increased gas solubility, resulting in a higher nucleation rate and finer cell structures. Conversely, temperature adjustments influence polymer viscosity and diffusion coefficients, thereby modulating cell growth and stabilization.
The resulting foam exhibits significantly enhanced properties. These include high energy return due to the elastic recovery of microcells, improved shock absorption from controlled compressibility, and reduced weight without compromising structural integrity. In addition, SCF foaming enables solvent-free processing, a critical advantage in reducing environmental impact and enhancing worker safety in manufacturing environments.
Drawing Parallels Between Coffee and Foam Processing
At a fundamental level, both decaffeination and foam fabrication rely on the dynamic phase behavior of SCFs to influence mass transfer, solubility, and diffusion phenomena. In coffee decaffeination, the target is the selective solubilization and removal of a low molecular weight compound embedded within a porous organic substrate. In footwear foaming, the goal is the uniform dispersion of gas within a polymer matrix to create a mechanically functional architecture.
The similarities extend to the process design philosophy. Both systems utilize phase equilibrium engineering to achieve selectivity and performance. In coffee, the operating envelope is tuned to extract caffeine without disturbing essential flavor compounds. In foaming, the same parameters are adjusted to achieve the desired balance between foam density, elasticity, and thermal stability.
Additionally, the recyclability and closed-loop nature of SCF systems in both cases offer significant sustainability advantages. Carbon dioxide, in particular, is non-toxic, readily available, and easily reclaimed, reducing both the carbon footprint and operational costs. This aligns with increasing environmental mandates in both the food and footwear sectors.
Opportunities for Cross-Disciplinary Innovation
The application of SCF technology in footwear is still in an evolutionary phase. However, leveraging process learnings from decades of SCF use in industries such as food and pharmaceuticals can significantly accelerate innovation. For instance, the understanding of co-solvent effects in caffeine extraction could inspire novel SCF-blended systems for foaming complex polymer blends or copolymers. Similarly, advances in pressure swing adsorption used to reclaim caffeine-laden CO2 streams could inform efficient gas recovery systems in foam manufacturing lines.
Furthermore, emerging techniques such as in situ spectroscopy and real-time pressure-temperature monitoring, common in the quality control of decaffeinated coffee, can be translated to inline diagnostics in foam production. These tools enable precise control over material properties, ensuring batch consistency and performance predictability.
Conclusion
The use of supercritical fluids in both coffee decaffeination and footwear foam engineering exemplifies the versatility of phase-engineered processing systems. Though the end applications diverge—flavor optimization in one case, mechanical performance in the other—the underlying scientific principles reveal a common toolkit of thermodynamic tuning, selective solvation, and microstructural control. As materials science continues to borrow from adjacent disciplines, the integration of SCF technology offers a compelling path forward for both sustainable manufacturing and high-performance product development.